Four crystal types of polylactic acid (PLA)
Polylactic acid (PLA) is an aliphatic polymer compound and a new type of degradable polymer material. It has many advantages that other polymer materials cannot match. First of all, its synthetic raw materials come from renewable crops, and the resin can be degraded into H2O and CO2 after use, which will not pollute the environment; secondly, PLA is non-toxic, non-irritating, has good biocompatibility, and is widely used in medical applications Sutures, drug sustained-release materials, etc.; moreover, PLA has good mechanical properties and can be widely used in industrial packaging, textiles, automobile industries and other fields. Due to these prominent features, PLA is increasingly attracting attention.
The polymerized monomer lactic acid of PLA has stereoisomerism, and there are two optical isomers of L-lactic acid and D-lactic acid, so polylactic acid also has left-handed poly L-lactic acid (PLLA), right-handed poly D-lactic acid (PDLA) and Racemic poly DL-lactic acid (PDLLA) three isomers. Among them, PLLA and PDLA are both crystalline polymers with a crystallinity of 40-60%; mesopolylactic acid (PDLLA) is a non-crystalline polymer that does not have crystallinity and is amorphous.
Compared with amorphous polylactic acid (PDLLA), the glass transition temperature Tg and melting point Tm of crystalline polylactic acid (PLLA/PDLA) are slightly higher. At the same time, crystalline polylactic acid has a higher modulus of elasticity and less variability. Smaller, the mechanical properties showed a larger gap. In addition to the difference in physical properties and mechanical properties, the crystallization of polylactic acid also has a certain impact on the degradation performance of polylactic acid, because the degradation of polylactic acid always starts from the non-crystalline region, and the molecules in the crystal region of crystalline polylactic acid The chains are arranged neatly and tightly, and are not easy to be damaged. At the same time, the crystallinity of polylactic acid also helps to improve the air permeability, heat resistance, and thermal stability of polylactic acid.
The crystal form mainly depends on heat treatment or processing technology. Under different external environment conditions, different types of crystal forms can be transformed into each other. The crystal form of polylactic acid is divided into four types: α crystal form, β crystal form, γ crystal form, and sc crystal form. This article mainly introduces four different types of crystal forms of crystalline polylactic acid.
α crystal form
The α crystal form was first discovered in PLLA by De Sanctis and Kovacs. Its structure is shown in the figure below. The unit cell parameters are a=1.07nm, b=0.645nm, c=2.87nm, and it is a quasi-orthorhombic crystal system. Later, Hoogsteen et al. found that the annealed PLLA fibers had the same crystal structure, but the unit cell structure parameters were slightly different. Usually, α crystals can be obtained by crystallization from melt, solution, or solution spinning at low drawing temperature and drawing rate.
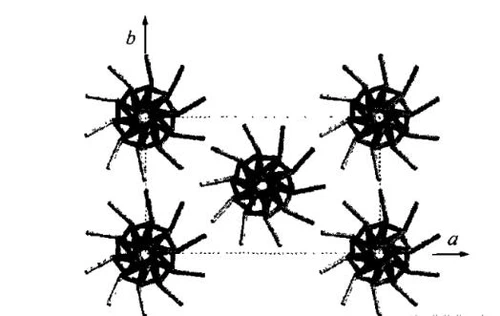
β crystal form
The β crystal form was discovered by Eling when he was studying the stretching of PLLA fibers, and it was mainly produced by mechanical stretching of the α crystal. Hoogsten proposed that the β crystal is an orthorhombic crystal system, and its unit cell parameters are a=1.03nm, b=1.82nm, and c=9.00nm. On this basis, Brizzolara proposed a broader molecular model of β crystals, thinking that there are two parallel molecular chains in the crystal structure unit. In recent years, Puggiali et al. found that the β crystal is a trigonal crystal system, with three triple-folded helices in the structural unit, and the parameters a=b=1.052nm, c=0.88nm. This structure enables PLLA to freely change the orientation of the upper and lower adjacent molecular chains under rapid crystallization conditions. The structure is shown in the figure below. From the above research results, it can be seen that the research on the β crystal form and unit cell parameters has not yet reached a consensus, and further research is needed.
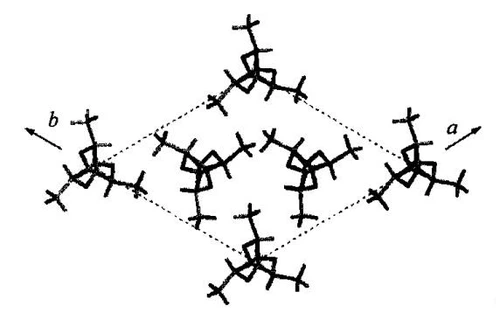
Gamma crystal form
The γ crystal form is obtained by the epitaxial growth of PLLA on hexamethylbenzene. The structure is very close to the β crystal molecular model proposed by Brizzolara, except that there are two antiparallel helices in the orthorhombic crystal system unit cell. The structure is shown in the figure below Shown, parameters a=0.995nm, b=0.625nm, c=0.88nm. Except for the γ-crystal obtained by hexamethylbenzene epitaxial growth, no other method has been found to obtain this crystal form.
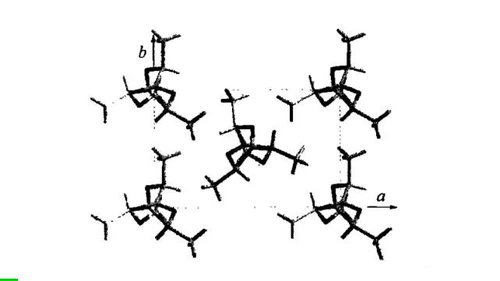
sc crystal form
The sc crystal form is a special crystal form of polylactic acid. It was first proposed by Ikada in 1987. It can be formed in the blend system of PLLA and PDLA. It is a stereocomplex crystal form. Compared with the pure PLLA and PDLA crystal forms alone, sc crystal materials have good heat resistance (melting point increased by about 50 °C) and chemical stability, so constructing sc crystals is an effective means to improve the comprehensive performance of PLA. Different proportions of PLLA and PDLA are mixed, and the appearance of the obtained sc crystal form is also different, as shown in the figure below.

Due to different crystallization conditions, pure PLLA/PDLA has three crystal forms of α, β and γ. The α crystal form is the most common and the most thermodynamically stable crystal form; the β crystal form can be obtained at a higher stretching ratio (10~20) and stretching temperature (180~200 °C), and the study found that the β crystal form PLA has Excellent impact resistance and heat resistance. Compared with α crystal, β crystal is unstable and its melting point is about 10 ℃ lower; γ crystal form is obtained by epitaxial growth of PLLA at about 140 ℃. The sc crystal form is a special crystal form produced by mixing PLLA and PDLA, and its melting point is greatly increased, up to 225°C.





